INTRODUCTION
Undifferentiated carcinoma of the pancreas with osteoclast-like giant cells (UCPOGC), first described by Juan Rosai in 1968, is a rare, highly malignant exocrine tumor [1]. UCPOGC is now classified by the World Health Organization as a rare variant of ductal adenocarcinoma, based on the epithelial origin of this tumor. In the early reports on this tumor it was suggested that they may have a more favorable prognosis compared with the usual ductal adenocarcinoma [2,3]. UCPOGC tends to present at the mean age of 60 years, and has the same sex distribution. Microscopic findings consist of atypical mononuclear round cells and abundant osteoclast-like multinucleated giant cells, thus, mimicking a giant cell tumor of the bone [2]. Here, we report a case of UCPOGC and describe its characteristics.
CASE
A 58-year-old man visited our outpatient clinic due to left upper quadrant pain for 3 consecutive months. The pain was continuous and it radiated to the back. The patient had weight loss of 8 kg over a 5-month period. He had type 2 diabetes mellitus, and was on medication for a duration of 5 months. No allergies or any significant family history was noted. On physical examination, he appeared to be chronically ill, with soft, non-tender abdomen. Vital signs were within normal range.
Complete blood counts and blood chemistries were within normal range. Carbohydrate antigen 19-9, alphafetoprotein, and carcinoembryonic cancer antigen were all normal. Ultrasonogram showed a 6.6 × 4.9 cm sized hypoechoic mass in the body of the pancreas.
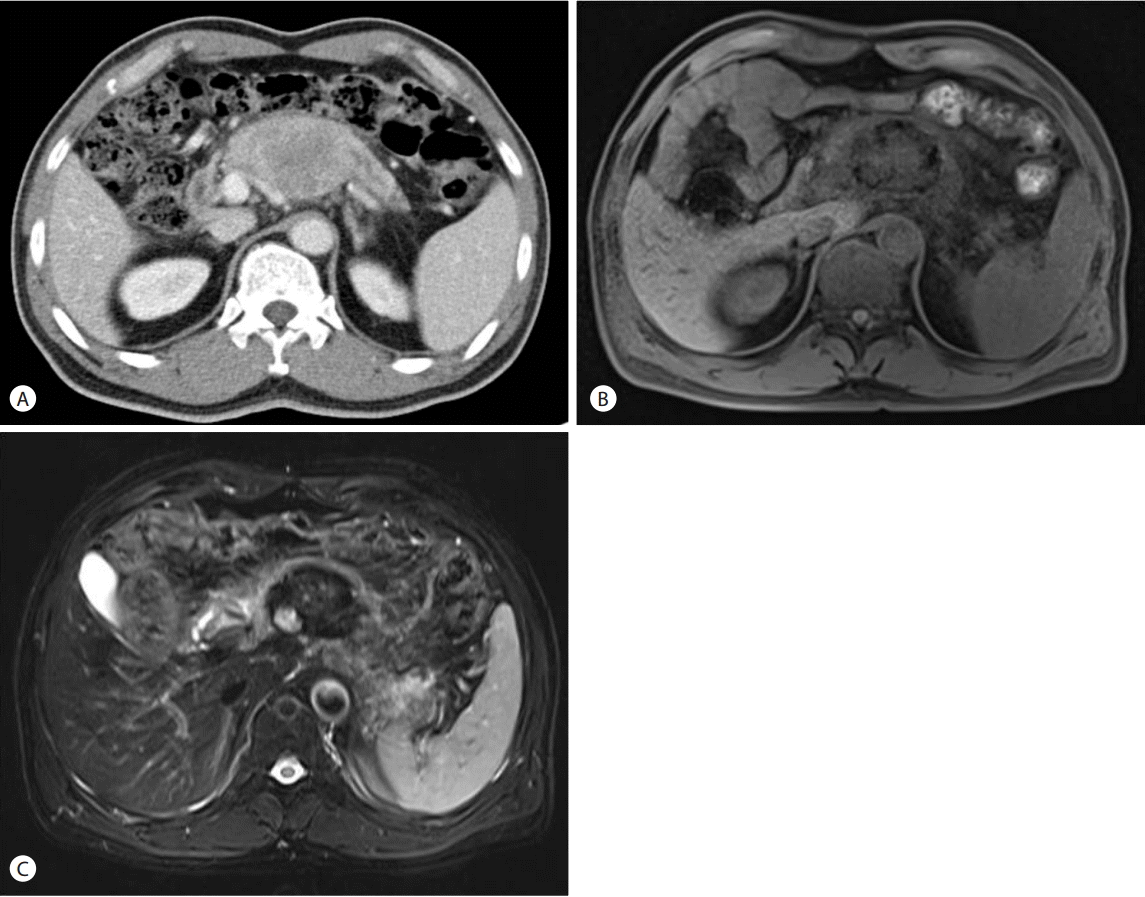
Abdomen CT and magnetic resonance imaging (MRI) showed a 5 × 5 cm sized heterogeneous pancreas body mass with an invasion of the splenic artery and vein, abutting the common hepatic artery and portal vein (Fig. 1). There were a few enlarged lymph nodes anterior to superior mesenteric vein and at the paraaortic and aortocaval area. Positron emission tomography-computed tomography (PET-CT) revealed no distant metastasis. Pancreatic cancer was suspected and EUS-guided fine needle aspiration (FNA) and biopsy with 19 gauge ProCore needle was done for obtaining tissue.
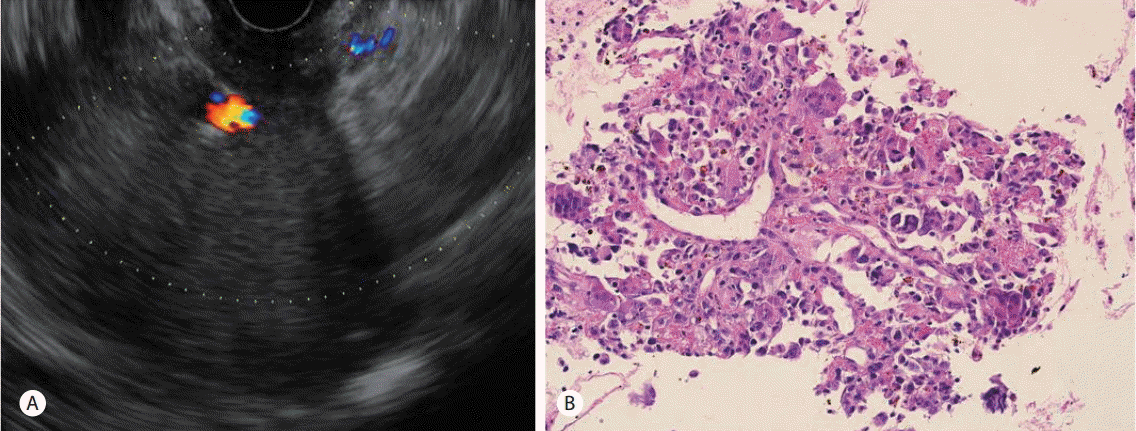
EUS showed a hypoechoic and heterogeneous mass on the pancreas body. The pathology of the specimen obtained by EUS-guided biopsy of the pancreas showed numerous neoplastic cells with a sheet-like arrangement, several multinucleated giant cells, and hemosiderin-pigments (Fig. 2).
Immunohistochemical stain showed cells positive for alpha-1-antitrypsin, vimentin, CD10 and beta-catenin, and negative for pancytokeratin and chromogranin. These findings suggested solitary pseudopapillary tumor (SPT) with marked degenerative changes.
Distal pancreatectomy and splenectomy were performed. Grossly, the resected mass was 5.4 × 4.8 cm in size and was composed of cystic and solid portions with necrosis and hemorrhage (Fig. 3A). The tumor arose from the pancreas tail and it extended beyond the pancreas. Resection margin involvement was absent, but lymphatic, intravascular, perivascular, and perineural invasion were noted.
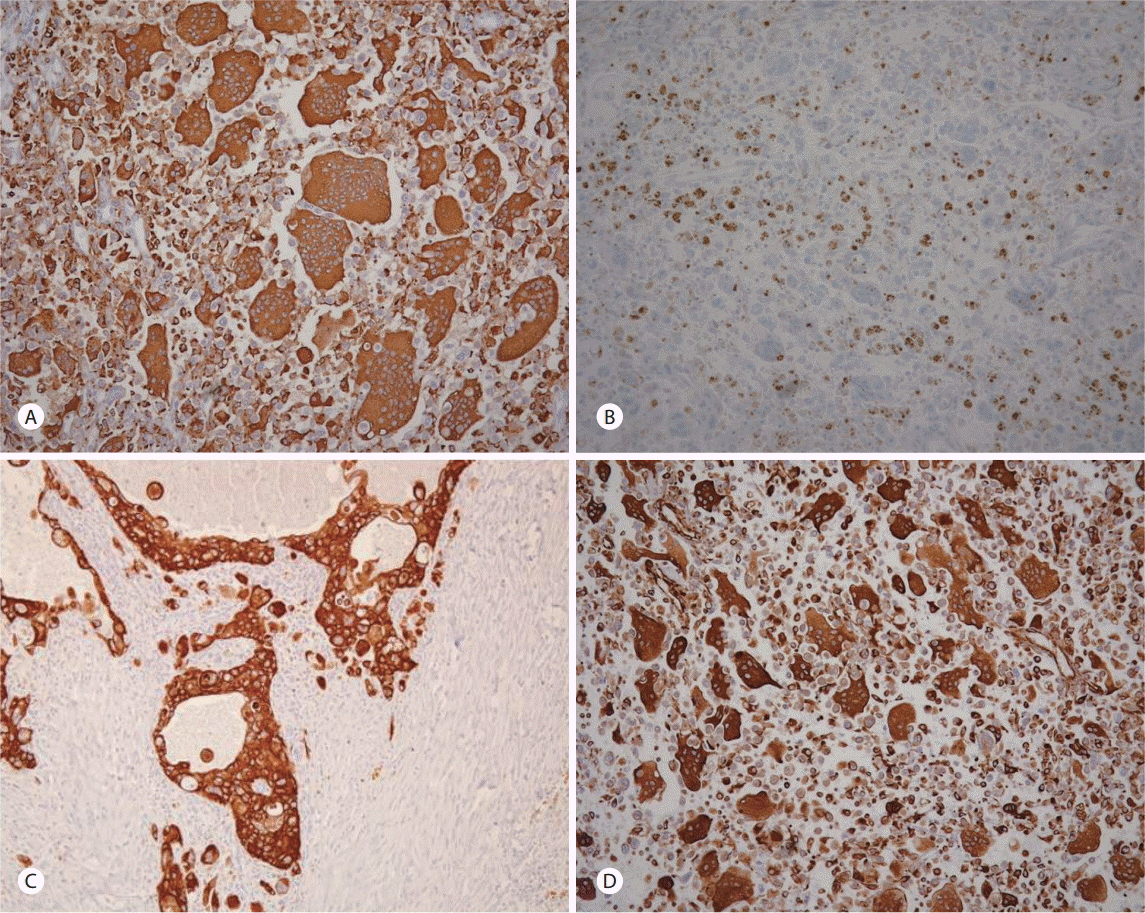
Histologic study showed tumor cell components consisting of atypical neoplastic mononuclear cells admixed with non-neoplastic osteoclast-like giant cells (OGCs) on hematoxylin and eosin stain (H&E) (Fig. 3B). The ductal adenocarcinoma component was also found in this case (Fig. 3C). On the immunohistochemical stain, OGCs were strongly positive for CD68, which are indicative of the histiocytic origin (Fig. 4A). Neoplastic mononuclear cells were positive for p53, but OGCs were negative for p53 (Fig. 4B). Pancytokeratin was also focally positive, which suggests the ductal cell origin of the undifferentiated carcinoma with OGCs (Fig. 4C). Vimentin was expressed in OGCs and neoplastic mononuclear cells (Fig. 4D). These findings were consistent with UCPOGC, and pathologic TNM staging was T3, N1, M0, stage IIB according to the TNM classification (7th edition) of the American Joint Committee on Cancer.
The patient received adjuvant chemotherapy with two cycles of 5-FU. However, metastatic cancer was detected on the soft tissue of the right thigh. The patient underwent palliative radiation therapy with two cycles of gemcitabine/erlotinib. Despite treatment, cancer progressed and the patient died 7 months after the initial diagnosis.
DISCUSSION
UCPOGC is a rare malignancy, which accounts for less than 1% of all pancreatic cancers [1]. It is an aggressive tumor that commonly invades adjacent organs. It is observed in patients with a mean age of 60 years but there is a wide age range, usually between 32 and 82 years. No gender predilection is associated with UCPOGC [2,4].
The main signs and symptoms are abdominal pain, weight loss, fatigue, anorexia, and palpable mass. The CT scan shows irregular cystic and solid tumor with heterogeneous enhancement by the contrast [5]. MRI findings of this tumor have not been established yet. Hur et al. [6] reported UCPOGC with low signal intensity on T1-weighted image and heterogeneous high signal intensity with multifocal cystic lesions on T2-weighted image. Other case reported heterogeneous high signal intensity within the mass on T1-weighted image that was thought to be a cystic hemorrhage [7]. Another case showed low signal intensity on T2-weighted image [8]. The explanation for these findings is not clear, but we hypothesize that various extents of hemorrhage and necrosis of this tumor contribute to diverse MRI findings. In present case, the mass showed low signal intensity on both T1- and T2-weighted MRI.
Tumor markers, CEA and CA 19-9 are less commonly elevated and not distinct. The diagnosis is usually made on surgical specimen. In some cases, EUS-FNA has shown to be effective, and an accurate diagnosis had been achieved by cytology test [2,9].
UCPOGC is classified by the World Health Organization (WHO) histologic classification system as a variant of ductal adenocarcinoma with two distinct cell lines: (1) highly pleomorphic mononuclear or multinucleated neoplastic cells and (2) large multinucleated non-neoplastic cells [2,10]. Usually, multinucleated, benign-appearing OGCs are dispersed among pleomorphic mononuclear or multinucleated cancer cells. Immunohistochemical findings showed that, OGCs were positive for histiocytic marker CD68 and were negative for the epithelial marker p53. Pleomorphic mononuclear cancer cells were positive for p53. These findings demonstrate an epithelial origin of UCPOGC [2].
The differential diagnosis of UCPOGC includes cystic lesions, such as serous cystic tumor, mucinous cystic neoplasm, intraductal papillary mucinous neoplasm, solid pseudopapillary neoplasm and ductal adenocarcinoma with cystic degeneration.
In our case, EUS-guided core biopsy was inadequate; findings revealed a sheet-like arrangement of tumor cells with a few multinucleated giant cells, suggestive of solid pseudopapillary tumor (SPT) with marked degenerative change; however, the surgical specimen showed p53-positive highly pleomorphic mononuclear neoplastic cells and abundant CD68-positive multinucleated OGCs on immunohistochemical stain. Characteristically, coexistence of adenocarcinoma component was also seen. Because the presence of non-neoplastic osteoclast-like giant cells is the histological hallmark of UCPOGC, we were confident to definitely conclude UCPOGC as a diagnosis [6].
The prognosis of UCPOGC is poor, but it is better than that of the usual ductal adenocarcinoma [2,3]. In a review of 32 cases of UCPOGC, the mean survival was 20.4 months after diagnosis or surgery. Most of the patients died of tumor within 1 year, but the other patient survived more than 15 years. UCPOGC have better prognosis and better response to adjuvant therapy compared with undifferentiated carcinoma without OGCs, because it is slower to metastasize and rarely metastasizes to the lymph nodes. In this context, WHO classification might have separated UCPOGC from undifferentiated carcinoma without OGCs [11,12].
Lung and liver metastasis were commonly reported. Peritoneum, bone, prostate and adrenal metastasis were also reported [12,13]. But, there is no report of soft tissue metastasis in UCPOGC patients until now. In our case, soft tissue metastasis was proven by biopsy.
Because UCPOGC is rare, it is difficult to determine an optimal treatment modality for this tumor. It is recommended to follow the protocols established for pancreatic ductal adenocarcinoma as a treatment modality. In general, surgical en-bloc resection is considered the first line of treatment and may prolong survival [5,6]. While, the efficacy of adjuvant treatments, including chemotherapy or radiotherapy, remains to be demonstrated. Recently, there was a case report of locally advanced UCPOGC patient treated by en-bloc surgical resection followed by adjuvant chemotherapy who survived more than 10-year without any evidence of recurrence [2]. However, further studies are needed to improve our understanding of UCPOGC.
In summary, a middle aged male with imaging studies showing a heterogeneous pancreatic mass was initially diagnosed as SPT with marked degenerative changes via EUS-guided biopsy. However, surgical specimen revealed two distinct cell lines—mononuclear neoplastic cells and large multinucleated osteoclast-like giant cells. The tumor was then diagnosed as UCPOGC.
Undifferentiated carcinoma of the pancreas with osteoclast-like giant cells is a rare, highly malignant pancreatic neoplasm. It is difficult to determine an optimal treatment modality for this tumor due to its rarity. Therefore, early diagnosis and complete resection could be only chance to cure this tumor. When irregular solid and cystic pancreatic mass observed on imaging test, as it did in our case, UCPOGC should be considered in the differential diagnosis.