INTRODUCTION
Intraductal papillary mucinous neoplasm (IPMN) of the pancreas is a cystic neoplasm originating from the epithelium of the pancreatic duct and is characterized by papillary intraductal growth of mucin-producing cells with variable degrees of cytological atypia, mucin hypersecretion, and cystic dilation of the involved pancreatic ducts [1,2]. IPMNs are classified as the main duct type, branch duct type, or mixed type according to the area of the involved ducts. The main duct type of IPMNs (MD-IPMNs) are frequently associated with a pancreatic duct obstruction and pancreatitis. Previous studies have reported an incidence of acute or chronic pancreatitis associated with MD-IPMNs ranging from 14 to 53% [3-5]. However, branch duct type of IPMNs (BD-IPMNs), which are usually small and indolent in their clinical course, are rarely associated with obstructive pancreatitis, and the incidence of acute or chronic pancreatitis is 5% [6]. Furthermore, BD-IPMNs are frequently found incidentally during routine radiological examinations and can be misdiagnosed as pseudocysts or other kinds of cystic tumors because of nonspecific imaging findings.
Although BD-IPMNs can also be the cause of acute or recurrent pancreatitis [7,8], the causal relationship is unclear, and published case reports are rare. Herein, we report a case of BD-IPMN leading to recurrent attacks of acute pancreatitis, which was initially thought to be pseudocysts complicated by idiopathic recurrent pancreatitis.
CASE
A 62-year-old woman was admitted to the hospital due to acute abdominal pain. The patient had suffered six episodes of recurrent acute pancreatitis during the past 5 years. She had been taking medications for diabetes mellitus, hypertension, and hypothyroidism. She did not smoke or drink alcohol.
The first acute pancreatitis episode occurred 5 years ago. Laboratory findings at that time included leukocyte count 19,700/mm3, hemoglobin 11.6 gm/dL, platelets 386,000/mm3, serum amylase 4,100 U/L, serum lipase 3,800 U/L, total protein 6.18 g/dL, albumin 3.85 g/dL, total bilirubin 0.7 mg/dL, aspartate transaminase 12 U/L, alanine transaminase 14 U/L, alkaline phosphatase 153 U/L, gamma glutamytranspeptidase 14 U/L, total cholesterol 150 mg/dL, triglycerides 100 mg/dL, calcium 7.0 mg/dL, and inorganic phosphorus 2.8 mg/dL. An abdominal computed tomography (CT) scan showed severe peripancreatic fat infiltration with fluid collection and pleural effusion (Fig. 1). Neither a cystic lesion nor ductal dilation was found at this time. No gallstones were detected on an ultrasonogram. A diagnosis of idiopathic pancreatitis was made. She improved with conservative treatment and was administered ursodeoxycolic acids under suspicion of microlithiasis as the cause of the pancreatitis.
She was readmitted 17 months later due to severe epigastric pain with elevated levels of serum amylase and lipase. An abdominal CT scan showed peripancreatic fat infiltration and two cystic lesions (2.0 and 1.5 cm) in the pancreatic body, which did not seem to communicate with the pancreatic duct (Fig. 2). The cystic lesions were considered pseudocysts because of the low-attenuation, unilocular cyst with accompanying acute pancreatitis. Her symptoms improved with conservative treatment. Two additional recurrent pancreatitis episodes developed 34 and 52 months after the first admission, but her symptoms improved with conservative treatment. The size of pancreatic cystic lesions decreased initially after the third pancreatitis episode but remained unchanged during further follow-up. A tubular shaped or grape-like cystic lesion without changes in size and shape in follow-up images even after full recovery of acute recurrent pancreatitis is highly suggestive of BD-IPMN. The abdominal CT scan revealed no typical BD-IPMN findings. However, we were strongly suspicious of BD-IPMN as the cause for the acute recurrent pancreatitis because of the lack of a change in the size and shape of the cystic lesions during the follow-up. Therefore, surgical resection or further evaluation, such as magnetic resonance cholangiopancreatography, endoscopic ultrasonography, and 18F-fluorodeoxyglucose-positron emission tomography combined with CT, were strongly recommended. However, the patient refused both surgical resection and further evaluation.
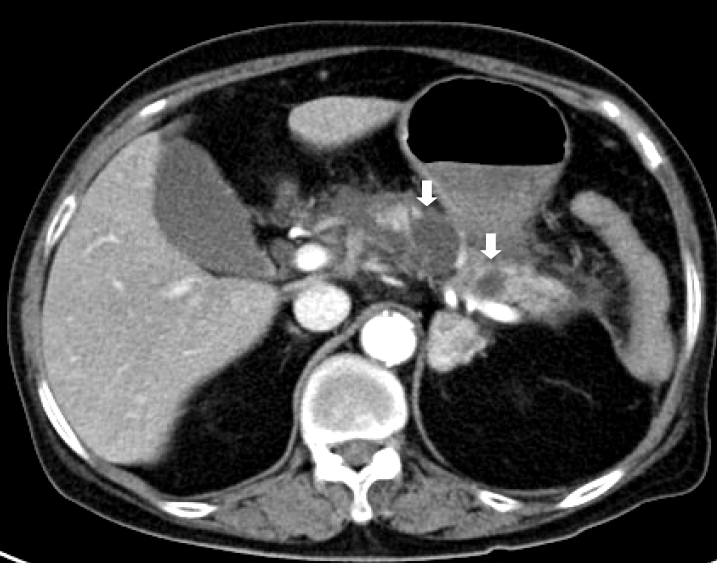
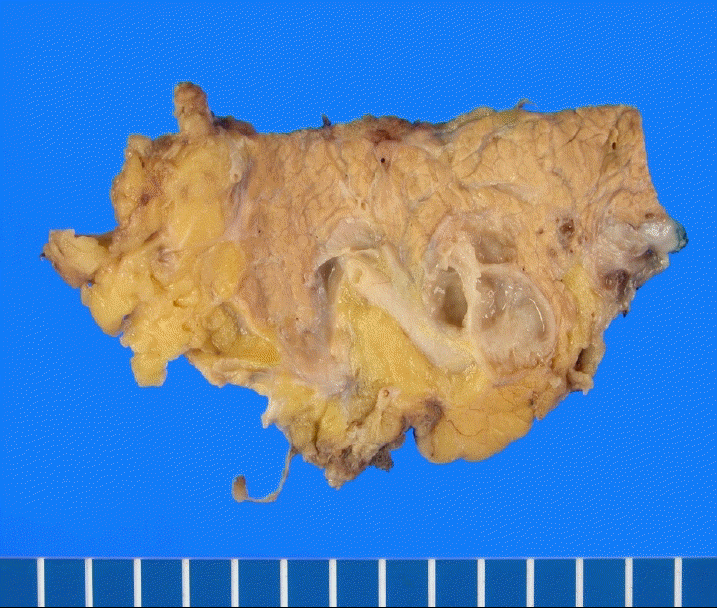
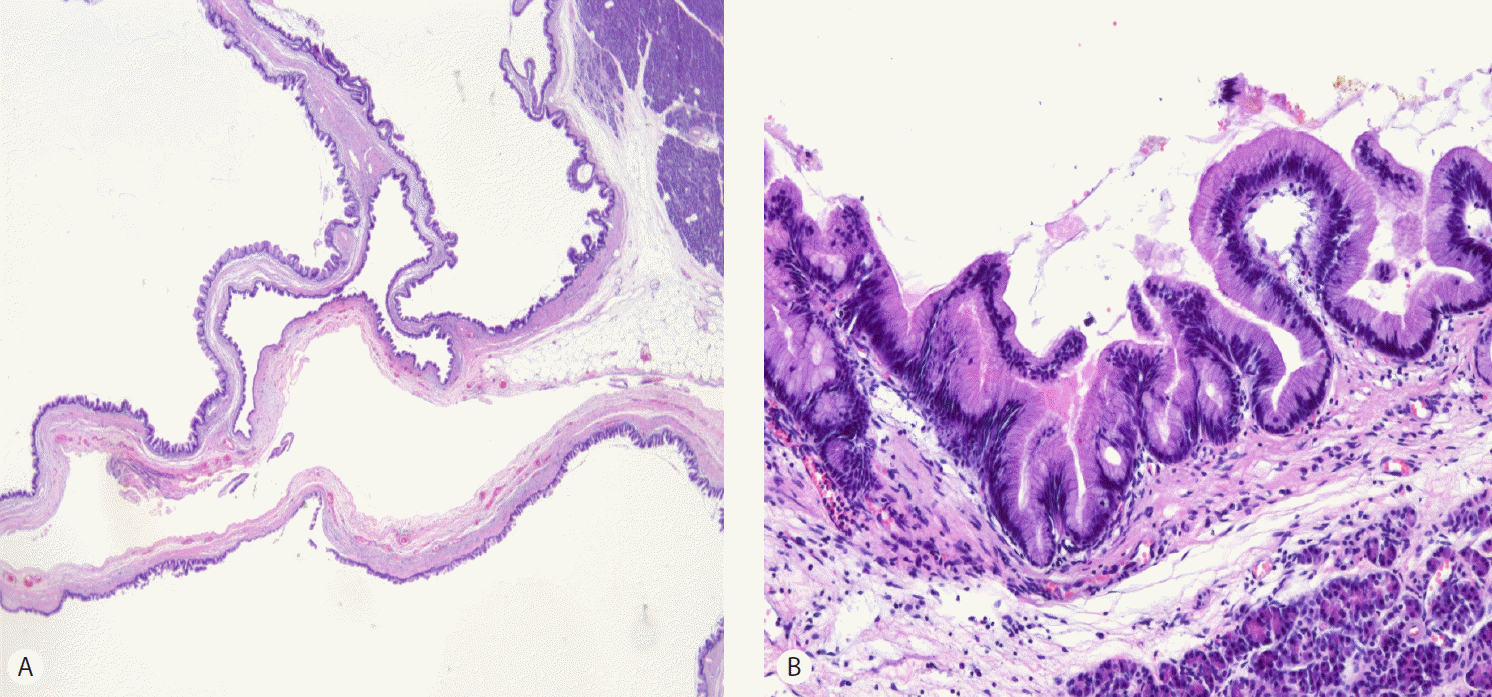
The patient was readmitted 4 months later due to a recurrent attack of acute pancreatitis. The cystic lesions had increased in size to 2.5 and 3.5 cm (Fig. 3). A distal pancreatectomy was performed after the pancreatitis improved under a recurrent pancreatitis diagnosis due to BD-IPMN. The gross findings of the resected specimen showed multiloculated cysts with septation (Fig. 4). The histological findings revealed two cystic lesions of 2.0×1.5 cm and 3.3×2.3 cm with multilobulation, and they were connected to each other in the pancreatic body (Fig. 5A). The cystic lesions were filled with mucin in the cytoplasm and had scattered elongated nuclei with moderate focal dysplasia (Fig. 5B). After surgery, the patient was discharged without complications and no recurrence of acute pancreatitis has been reported during the follow-up.
DISCUSSION
IPMN of the pancreas is a rare pancreatic tumor originating from the epithelium of the pancreatic duct that is characterized by mucin production. In most IPMN cases, hypersecretion of mucin leads to cystic dilatation of the involved ducts. IPMNs are categorized into the main duct type, the branch duct type, and the mixed type based on imaging studies or histology. The majority of the BD-IPMNs are small cystic masses without dilation of the main duct, whereas MD-IPMNs are highly suggested when dilatation of main pancreatic duct is >1 cm.
MD-IPMNs and BD-IPMNs have significant differences in their clinical course and malignant potential. BD-IPMNs are typically smaller, less complex (less papillary), and less malignant compared to those of MD-IPMNs, which explains why many BD-IPMNs have been successfully managed conservatively. The reported incidence rate of cancer in the main and branch ducts of IPMNs are 57-92% and 6-46%, respectively [9,10]. BD-IPMNs are asymptomatic in 86% of cases, and abdominal pain is the most frequent symptom in symptomatic cases. The mechanism of pancreatitis in BD-IPMNs seems to be similar to that of MDIPMNs. Viscous mucinous secretions from IPMNs may obstruct the main pancreatic duct or branch duct, leading to elevated pancreatic duct pressure and subsequent pancreatitis [11].
Concomitant BD-IPMNs associated with acute pancreatitis are frequently confused with pseudocysts. One case report on IPMNs presented that the initial diagnosis was a pseudocyst, and a pseudocyst-jejunal anastomosis was performed [12]. In our case, we initially considered that the cystic lesions were pseudocysts complicated by acute pancreatitis because the lesions were not present at the time of the first attack but appeared after the second attack of pancreatitis. The relatively long interval of >1 year between the first and second pancreatitis attacks made the diagnosis more difficult. We thought that the initial pancreatitis attack may have been caused by pre-existing small BD-IPMNs, which were masked on the CT scan, by severe inflammation during the first attack of pancreatitis. Clues leading to the diagnosis of BD-IPMN in our case were as follows: (1) no obvious potential cause for recurrent pancreatitis; (2) the relatively small cystic lesions did not disappear even after improvement in pancreatitis; and (3) the size and position of the cysts were unchanged during a long-term follow-up. Some fluctuations in the size of the BD-IPMNs were likely related with pancreatitis, which can temporarily increase the size of BD-IPMNs by blocking the pancreatic duct with mucin plugs.
IPMNs are generally slowly growing tumors with a good prognosis. A recent international consensus guideline recommended nonsurgical management with close observation of IPMNs that arise from branch ducts and that fail to meet certain “high-risk” criteria, including causes attributable to the cyst (such as pancreatitis), cyst size of at least 30 mm, presence of intramural nodules, or cyst fluid cytology suspicious or positive for malignancy [13]. BD-IPMNs appear to be a risk factor for pancreatic cancer and careful observation is recommended [14,15]. Although the BD-IPMN in our case was a very slow growing tumor over 5 years, there was a high possibility of progression to malignancy if surgical resection was not performed because the pathological findings showed moderate dysplasia.
In conclusion, BD-IPMNs should be considered as a cause of recurrent pancreatitis of unknown etiology. The presence of pancreatic cystic lesions in patients with idiopathic pancreatitis suggests the possibility of BD-IPMNs as the cause of the pancreatitis, particularly when the cysts are small and unchanged during follow-up.